|
So, what drives DNA strands together? Entropy and enthalpy, of course. (The H-bonds do contribute *something*: GC base pairs with three H-bonds are harder to melt than AT pairs with two.) of G-C or A-T is of the same energy (sometimes even less) than the hydrogen bonds these same groups would form with water if the DNA were single-stranded. Hydrogen bonding between the amines, carbonyl oxygens, etc. For an interaction to stabilize the duplex, the interaction between bases must be stronger than the interaction of the bases with water (if bases are not paired with one another in a duplex, then they are surrounded by water). This cloud of counterions lowers the effective charge density and relieves the repulsion between the strands.Īs for the effect of salt on hydrogen bonds, you have to realize that the hydrogen bonds formed between bases in duplex DNA contribute little to the stability of the duplex. Cations added to the solution form a "cloud" of positive charges around the DNA. Each strand of DNA has an enormous charge density (charge per unit volume), so the two strands tend to push each other apart. Q: Why does NaCl increase the stability of DNA duplexes, although you might expect salts to interfere with hydrogen bonds, rather than strengthen them?Ī: The Na+ neutralizes the charge. As e goes down, F goes up and *BANG*, anion and cation form an ion pair and promptly swoon out of solution. Adding organic solvent *decreases* the dielectric constant of the solution. Where Q is the charge on each ion and r is the distance between them. Water has a high dielectric constant (e), which from Coulomb's Law tells us that the electrostatic force (F) between two ions of opposite charge is very low in water: In dilute aqueous solution, DNA and counterions like Na+ and Mg++ are more or less in the free ion form rather than the ion pair form (that is, they are surrounded by one or more layers of water molecules). For a precipitation, you're interested in forming ion pairs between the polyanion (DNA) and the cation (Na+, Mg++, spermidine, protamine, etc). Q: What is the role of ethanol in precipitating DNA from aqueous solutions?Ī: The purpose of adding salts is to neutralize the charge on the sugar-phosphate backbone of the DNA, but ethanol's task is a little more complex than "removing" the water. A thorough discussion was presented by Scott Keeney, of the Biochemistry and Molecular Biology Department, University of California Berkeley I have shortened the opening questions and extracted his answer from the newsgroup and present them below for your convenience. The anions and cations form ion pairs and this results in precipitation of DNA.Ī discussion on this topic was posted to the "-reagnts" bulletin board in July 1992. Adding ethanol decreases the dielectric constant of the solution. The DNA and counterions are more or less in the free ion form rather than the ion pair form in dilute aqueous solution, and are surrounded by one or more layers of water molecules. 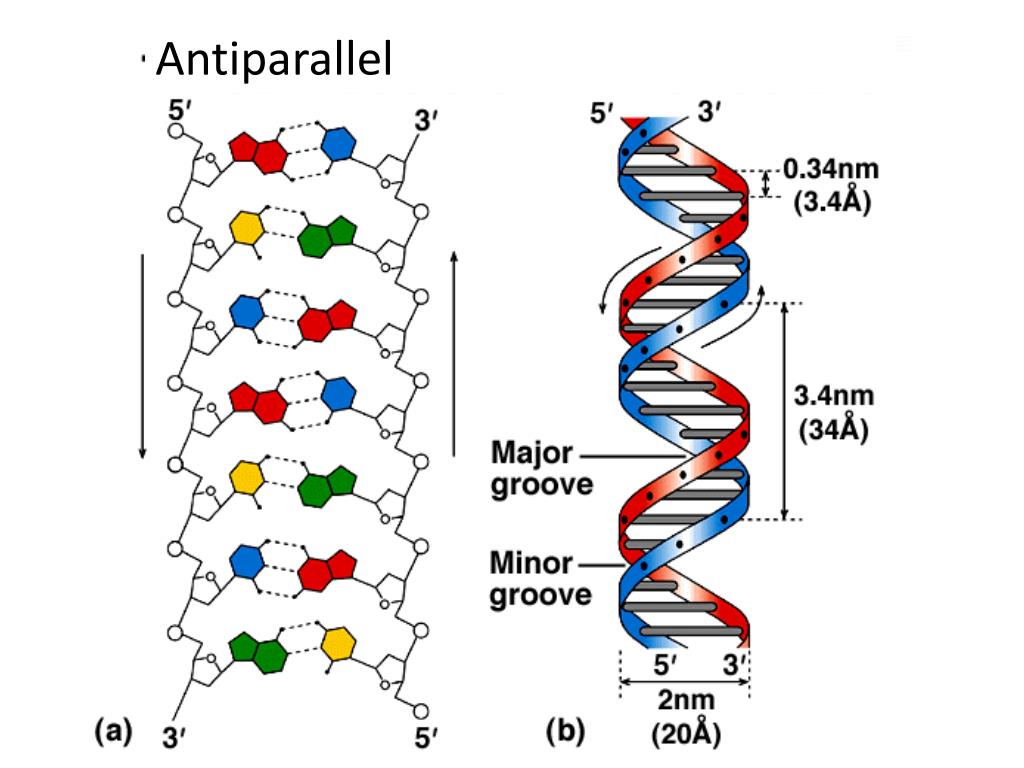 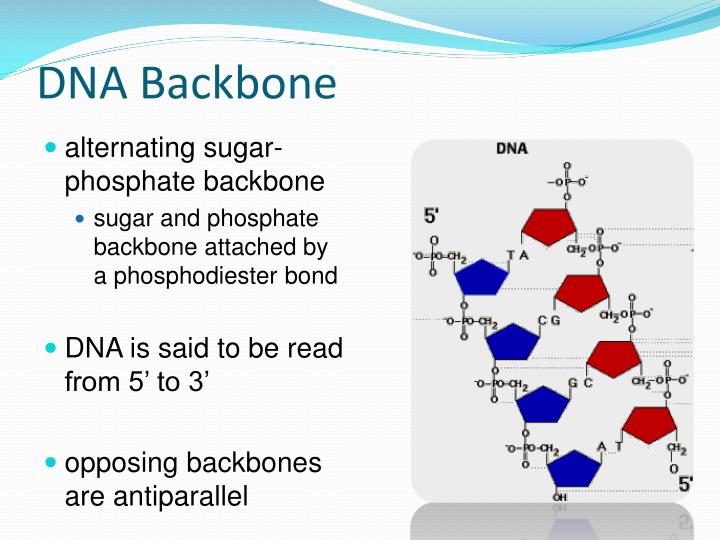
For a precipitation, you need to form ion pairs between the polyanion (DNA) and the cation (usually sodium or ammonium). The purpose of adding salts is to neutralize the charge on the sugar-phosphate backbone of the DNA.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
